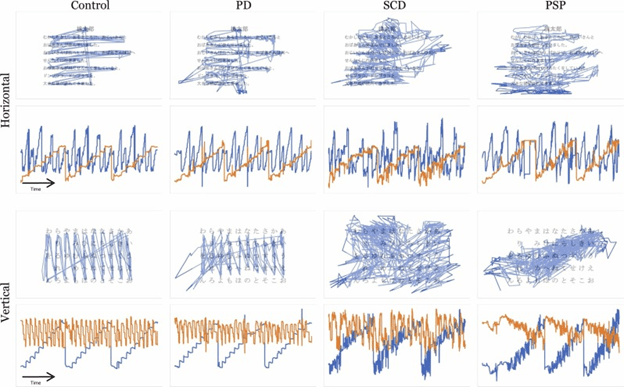
A very active area of research right now is how various imaging techniques (“scans” in English) can and cannot assist in distinguishing the atypical Parkinsonian disorders from other conditions and from one another. Yes, this is important for clinical care and counseling. But even more important right now is that until we have specific treatments for these diseases, we need accurate diagnosis in living people. This is important for laboratory researchers who want to know the true diagnosis of the patient who supplied a fluid sample, and to the designers of clinical trials who want to make sure the patients in their trials have the disease for which the treatment was designed.
Dr. Jennifer L. Whitwell, a radiology researcher at the Mayo Clinic in Rochester, MN has just published a very helpful review of that topic in Current Opinion in Neurology. It gets pretty technical, but here are the takeaways with, of course, my own editorial contributions:
- Measurement of atrophy by MRI:
- The magnetic resonance Parkinsonism index (MRPI), especially its updated version, the MRPI 2.0, gives excellent differentiation of PSP from non-PSP, with an area under the receiver operating curve of 0.98. (That statistic is 1.00 for perfect accuracy and 0.50 for no better than a flip of a coin. For a bit more explanation of the AUROC and a graph, see this post from last year.)
- Routine MRI can also be used to compare the various PSP variants with regard to atrophy of specific brain structures. Atrophy of the brainstem is worse in PSP-Richardson syndrome, PSP-CBS and PSP-frontal than in the others. This information could be useful in treatment trial design because some PSP variants progress faster than others. So, a trial of a disease-slowing treatment could potentially determine which patients have which variants and adjust the statistical analysis of the treatment outcomes accordingly.
- MRI shows that atrophy in PSP-Richardson’s syndrome starts in the midbrain, (the upper part of the brainstem where the substantia nigra and the vertical gaze centers reside), followed by a succession of other areas of the brainstem, cerebellum and basal ganglia, before finally reaching the cerebral cortex. But two PSP subtypes involving relatively more cortical function (the frontal behavioral type and the corticobasal syndrome type) spread into the frontal cortex much earlier in the process, although like PSP-RS, they start in the midbrain. These observations could help guide designers of other imaging-based diagnostic tests for PSP.
- Measurement of metabolism by fluorodeoxyglucose positron emission tomography (FDG PET): The pattern of reduced brain tissue metabolic activity in PSP can be distinguished from normal with an AUROC of 0.99. Distinguishing PSP from corticobasal syndrome, multiple system atrophy and Parkinson’s disease is more difficult but still useful at 0.90.
- Measurement of tau deposition by PET: A PET technique that reveals the location of abnormal tau in Alzheimer’s is already in standard clinical use for AD, but it doesn’t work well for other tauopathies. Two techniques designed for PSP are expected to enter pivotal clinical trials in the next few months. In small studies, the two ligands called PI-2620 and APN-1607 (formerly called PM-PBB3) have shown good results in distinguishing PSP from the other tau disorders and Parkinsonian disorders. (A PET ligand is the chemical injected intravenously that sticks to the brain chemical of interest, allowing its location to be mapped.) But these two ligands can occasionally give conflicting results when given to the same patient, so the AUROCs from the upcoming trials are eagerly awaited.
- Measurement of iron deposition by MRI: This technique, called quantitative susceptibility mapping (QSM), is not part of a routine MRI, but it can be performed with the same machine. Its PSP-diagnostic results are not quite as accurate of the ones above, with an AUROC of 0.83 for distinguishing PSP-Richardson’s syndrome from Parkinson’s disease. I assume the AUROCs for other kinds of PSP, especially PSP-Parkinsonism, are worse, but work continues on this technique.
- Functional MRI: The spread through the brain of abnormal tau, and with it the damage of PSP, proceeds along “functional networks.” That means that after first affecting the substantia nigra in the midbrain, the damage proceeds to areas most closely yoked to it in terms of simultaneous electrical activity. Those physiological relationships have been delineated by “functional MRI” technique, where a person is given various mental or motor tasks to perform while in the MRI machine. The MRI software is set to measure not physical or chemical structure as for routine MRI, but blood flow, which correlates exquisitely with brain tissue electrical activity. These observations could potentially allow researchers to assess the effects of experimental drugs on the immediate or longer-term pattern of brain cell activation in people with PSP.
Exciting developments all, and I apologize for nerding out on you yet again. But as one of this blog’s commenters said a few years ago, “Thanks, Dr. Golbe, for respecting our intelligence.” Still, I’ll try softer stuff next time.