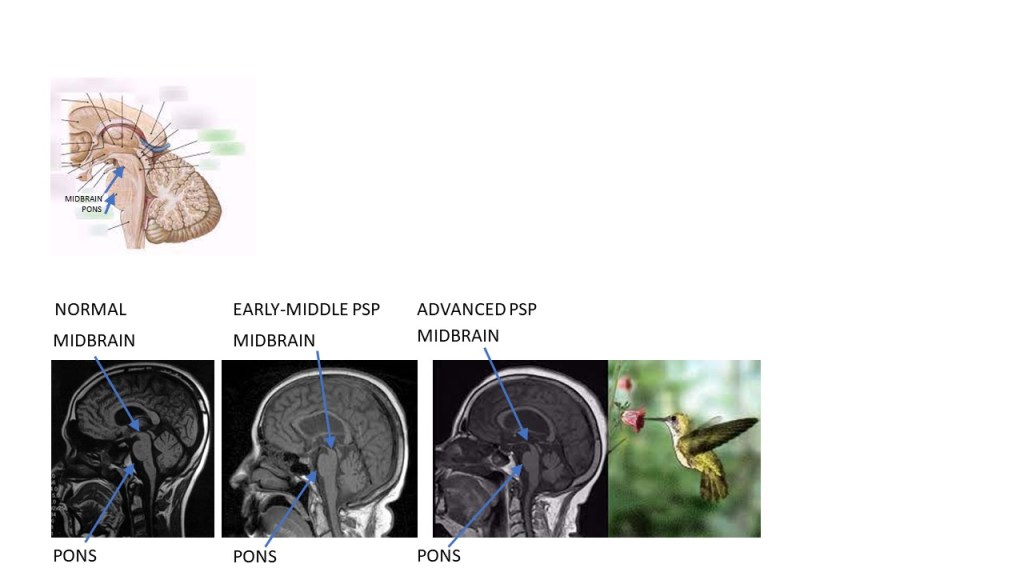
Back to my reports on the new PSP research presented at the International Parkinsonism and Movement Disorder Society conference in Copenhagen last month. Last week I started with the treatment-related things. Today it’s five presentations on diagnostic tests other than imaging. As before, my editorial comments appear in italics.
Machine learning classifies Parkinson’s disease and progressive supranuclear palsy on saccade, pupil, and blink measures during a naturalistic free-viewing task
D. Brien, H. Riek, R. Yep, J. Huang, B. Coe, B. White, M. Habibi, D. Grimes, M. Jog, A. Lang, C. Marras, M. Masellis, P. Mclaughlin, A. Peltsch, A. Roberts, B. Tan, D. Beaton, W. Lou, E. Finger, A. Frank, D. Tang-Wai, C. Tartaglia, S. Black, R. Swartz, W. Oertel, D. Munoz (Kingston, Canada)
The researchers tracked eye movements using artificial intelligence software in 120 people with Parkinson’s disease, 8 with PSP, and 97 controls viewing a 10-minute movie. They told the software the diagnosis and allowed it to create a profile of what sorts of eye movements correlated with which disease. The result was 87% accuracy in distinguishing PD from PSP in a separate group of patients. (The percentage refers to the area under the receiver operating curve – see my post on that statistical technique if you like).
This technique is no more accurate than an exam by a skilled movement disorders specialist, but is potentially much easier to obtain and pay for. Plus, it’s non-invasive.
Utilizing speech analysis to differentiate progressive supranuclear palsy from Parkinson disease
K. Kang, A. Nunes, M. Sharma, A. Hall, R. Mishra, J. Casado, R. Cole, G. Barchard, A. Vaziri, A. Wills, A. Pantelyat (Baltimore, USA)
This is similar to the previous presentation in that it uses computerized analysis of patients’ fine movements to differentiate PSP from PD. In this case, researchers analyzed multiple features of speech during passage reading, counting and a sustained “aah” sound. They found multiple clear differences between PSP and PD in the average measurements of many features of speech but did not report areas under the receiver operating curve, so we don’t know how useful the measurements are at the individual level. The researchers conclude that automated analysis is a feasible and non-invasive way to differentiate PSP from PD.
We’ve long known that the speech of PSP differs from that of PD, and an experienced neurologist can easily distinguish the two over the phone. But this may be the first time the differences have been quantified so precisely. This small study (only 11 patients with PSP, 10 with PD) should be replicated and its value at the individual level measured. It all goes well, this could become a smartphone app.
Cognitive interference in postural control as a diagnostic and prognostic biomarker in Parkinsonian disorders.
R. Lloyd, C. Fearon, R. Reilly (Dublin, Ireland)
It’s well known that in PSP and PD, the balance problem is worse when the person has to concentrate on a cognitive task at the same time. These researchers presented their plans for using that phenomenon to distinguish between the two diseases. They will use an automated platform for balance assessment and the popular Stroop color-word test for cognition, with the patients attached to an electroencephalogram (EEG; brain wave recording) machine. They expect that the cognitive task will aggravate forward/backward sway in PD and side-to-side sway in PSP, and that corresponding EEG changes will occur. There’s no description of the results as of the conference’s submission date, nor on my PubMed search today.
While this elaborate testing set-up would not be practical for real world use, it may be relatively easy to have patients walk down a clinical corridor, with appropriate safety measures, with and without simultaneous performance of a cognitive task. The neurologist could eyeball the result. Of course, that simplified version would require its own validation procedure before being recommended.
Remote monitoring of physical activity in progressive supranuclear palsy (PSP) using wearable sensors
A.-M. Wills, R. Mishra, M. Sharma, AJ. Hall, J. Casado, R. Cole, A. Vaziri, A. Pantelyat (Boston, USA)
This project tested a wearable motion sensor that has been proven in PD for its value in PSP. Eleven patients with PSP wore the “PAMSys” device to measure overall activity, gait, and posture. The results were compared to a version of the PSP Rating Scale modified for tele-neurology use. The device was able to document disease progression over the 12-month period of observation.
Although little actual analysis of the data was presented in the abstract available. But this result does show that objective measurements of the ability of patients with PSP to move around may not have to rely on snapshots in time every 3 months for clinical trials or less often for regular clinical care. This avoids the technical difficulties in performing evaluations by video and the subjectivity of reports from patients or caregivers. I’d expect such measurements to start appearing as secondary outcome measures in clinical trials very soon.
Posturography as an objective measure of disease progression and prognostication in progressive supranuclear palsy
G. Nuebling, S. Katzdobler, J. Levin, G. Höglinger, S. Lorenzl (Munich, Germany)
As an add-on to a 12-month drug trial for PSP from a decade ago, these researchers used an automated platform to measure the degree of sway of 44 patients with PSP with eyes open and then with eyes closed. The delay since the data were gathered isn’t a result of procrastination. It was to be able to assess the sway data as a predictor of long-term survival. The result was that the data correlated very well with survival. But the precision of the test for sole use as an outcome measure in 12-month neuroprotection trials was insufficient. The result with eyes open was a better predictor than with eyes closed (hazard ratios 1.098 vs 1.001), but the sizes of that effect were small and the difference between them is not statistically significant.
We know a lot about factors that affect long-term disability and survival in PSP, but not in an easily applied, quantitative way that could be used in clinical care. This test could provide that if the apparatus can be modified to require less space in a clinic.