I wish I could report on some breakthrough in the treatment or prevention of PSP, but that hasn’t happened yet. But what I can do is to report on a major advance in understanding an “upstream” step in the process that’s killing brain cells in PSP. The abnormal tau molecule in PSP has now been imaged.
The prevailing, and probably correct, theory is that brain cells are damaged somehow by tau protein that has folded upon itself, causes its healthy sibling tau molecules to similarly misfold via a templating process. The tau then forms stacks like checkers, called “fibrils,” which are toxic to the cells. Tau doesn’t normally have any folding pattern at all – if it’s not attached to the cells’ internal skeleton, doing it’s normal job, it’s floating around in the cytoplasm (the cells’ internal fluid) like overcooked spaghetti in boiling water. We’ve known for decades that through the electron microscope, the fibrils of PSP look different from those of Alzheimer’s, which in turn are different from those of corticobasal degeneration, which are different from those of Pick’s disease, and so on. We’ve hypothesized that this is because the pattern of misfolding determines the geometry of the stacks of tau, and those stacks determine which cells are attacked, and that determines the patient’s symptoms. But we had no details.
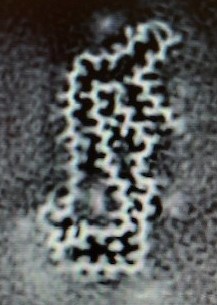
Well, now we do, thanks to a new lab technique called “cryo-electron microscopy” or “cryo-EM.” That’s where the sample is imaged at a temperature cold enough to keep it from wiggling around as much, allowing a far greater visual resolution than was possible using other methods. It still does wiggle a bit, which is why the technique starts by making a video of a tau molecule and then computer-averages the image. The resulting still photo shows the protein molecule’s pattern of loops and folds; even the individual amino acids are visible as fuzzy bumps on the chain. The resolution of the images was 2.7 Angstroms, about the same as the diameter of a water molecule.
Over the past couple of years, researchers applied cryo-EM to tau in a few tauopathies including AD, CBD and Pick’s disease. Only now has PSP been studied by this technology, along with a bunch of much less common tauopathies. The new paper is from an international team led by Michel Goedert, Sjors H. W. Scheres, Yang Shi and Wenjuan Zhang, all of Cambridge University. It was published in Nature, probably the world’s most prestigious and selective biomedical journal.
They imaged tau from 11 diseases, a number large enough to justify a classification system using a few different features such as the number of layered folds and hairpin turns. They found that the folding patterns of the various PSP subtypes were identical except for one case of a rare type, PSP-F, a variant with disproproportionate frontal lobe behavior and cognitive problems. Under the new classification system based on tau folding, the tauopathy most similar to PSP was globular glial tauopathy, a rare cause of dementia diagnosable only at autopsy.
They found the folding pattern of Alzheimer’s tau to be identical to that of primary age-related tauopathy (PART), familial British dementia (FBD) and familial Danish dementia (FDD). The pattern in chronic traumatic encephalopathy (CTE) was similar but not identical to that. Corticobasal degeneration (CBD) was similar to argyrophilic grain disease (AGD) and aging-related tau astrogliopathy (ARTAG). Finally there was Pick’s disease, which was similar to none of the other ten.

Why is all this important? I’ll let the authors themselves explain:
SHI Y ET AL. NATURE 2021
“The presence of a specific tau fold in a given disease is consistent with its formation in a small number of brain cells, followed by the prion-like like spreading of tau inclusions. This may underlie the temporo-spatial staging of disease. Knowledge of the tau folds in the different diseases provides a framework for studying tauopathies that will lead to a better understanding of disease pathogenesis. At a diagnostic level, our findings will inform ongoing efforts to develop more specific and sensitive tau biomarkers.”
By “temporo-spatial spreading,” they mean where in the brain the damage spreads and how fast. So the next big step is to figure out just how the molecular structure presented on the surface of each kind of misfolded tau interacts with healthy brain structures. Once we do that, we can find a monkey wrench to throw into that process – one monkey wrench for PSP and globular glial tauopathy; another for AD and its pals PART, FBD and FDD and possibly CTE; one for Pick’s; and yet another for CBD, AGD and ARTAG.
I’ll try to keep you updated on this, and soon I’ll opine on the collateral question of whether this new work casts doubt on our long-held position that PSP, as a “pure tauopathy,” is a good test-bed for all of the tauopathies, including AD.